A new gene inhalable therapy is generating interest in the scientific community after receiving an accelerated designation from the FDA. This experimental approach allows treatment to be delivered directly into the lungs. Early trials show promising results, marking a possible advance in developing more precise and less invasive treatments.
How the gene inhalable therapy against lung cancer works

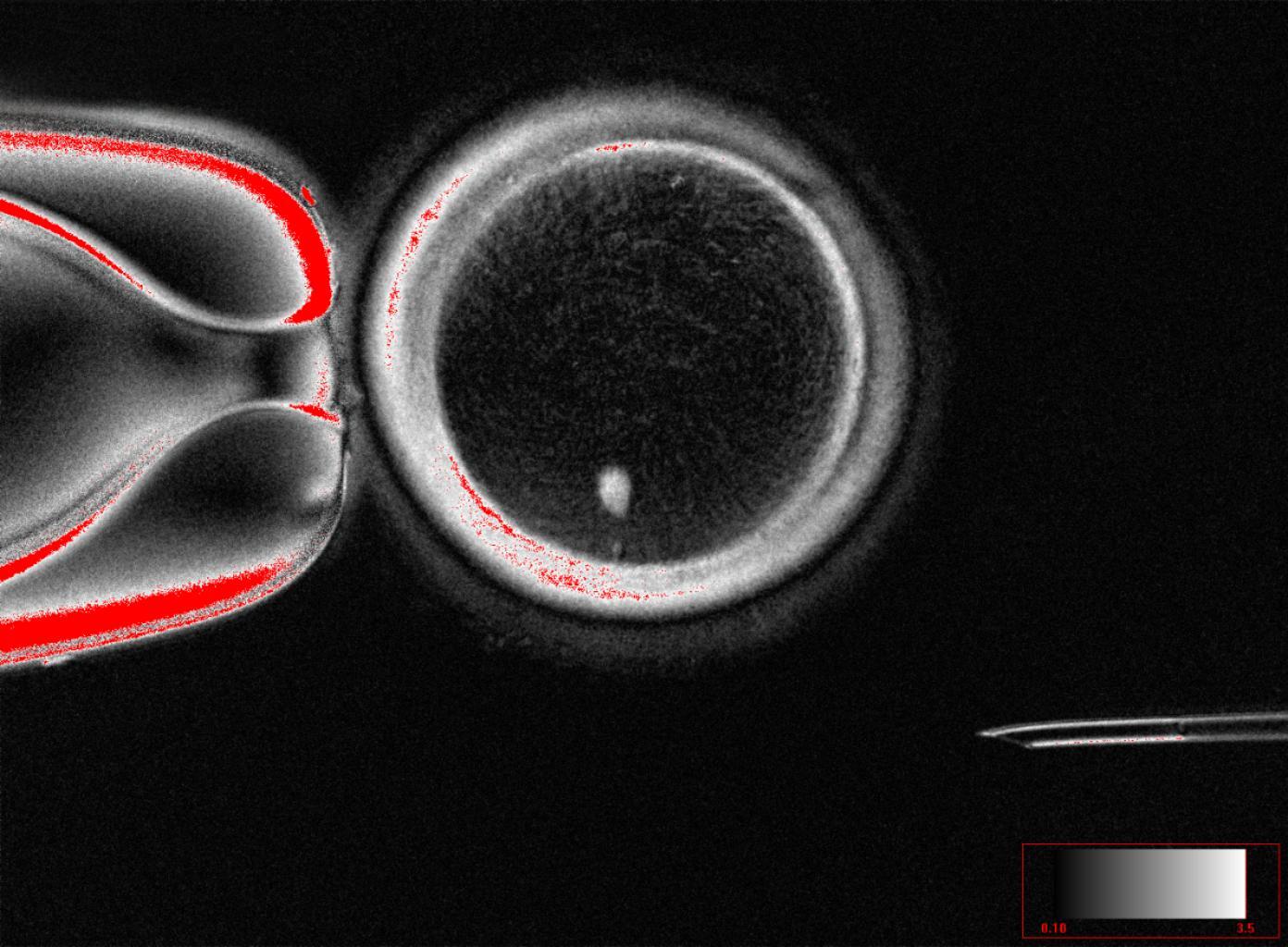
The gene inhalable therapy represents an innovative approach designed to act directly in the lungs, the organ affected by the tumor. Unlike traditional treatments, this method uses a nebulization, similar to that of some common respiratory medications.
The treatment contains a modified viral vector, a virus designed to be safe and incapable of causing disease. Its function is to transport specific genes into lung cells. These genes help activate the immune system in the area where the tumor is located.
Once inhaled, the viral vector reaches the lung tissue and delivers genetic instructions that stimulate the production of immunostimulatory proteins. These proteins help the immune system recognize cancer cells as a threat.
In many cases, cancer manages to evade the body’s defenses. This therapy seeks to reverse that process by strengthening the local immune response. The goal is for the body itself to actively participate in controlling or eliminating the tumor.
Another important aspect is that the treatment acts locally. This means its action is concentrated in the lungs, reducing exposure to the rest of the body. This approach could decrease some side effects associated with traditional systemic therapies, such as chemotherapy.
Although the concept is complex, the principle is clear: using genetic engineering to help the immune system fight cancer more precisely.
Preliminary results and available clinical evidence

The first clinical trials of this therapy have shown encouraging results in patients with advanced lung cancer. According to reported information, some participants experienced a reduction in tumor size, while others showed stabilization of the disease.
These results were presented at specialized scientific meetings, where researchers highlighted the treatment’s potential. The observed response suggests that targeted activation of the immune system can have a real effect on the tumor.
However, it is important to understand that these trials are in early phases. The main objective at this stage is to evaluate safety and determine the appropriate dose, in addition to observing possible clinical benefits.
Not all patients respond in the same way. This is common with innovative therapies, especially in early research stages. Scientists continue collecting data to better understand which patients may benefit the most.
The accelerated designation granted by the FDA reflects that the therapy has potential, but it does not mean it is approved for general use. Larger studies are still required to confirm its effectiveness and long-term safety.
Potential advantages compared to traditional treatments

One of the main benefits of this therapy is its non-invasive administration method. Instead of requiring surgery or complex intravenous infusions, the treatment can be delivered through inhalation.
This approach allows the medication to reach the tumor site directly. This precision could improve treatment effectiveness and reduce the impact on healthy tissues. In comparison, traditional treatments often affect both cancerous and normal cells.
Another potential advantage is the reduction of some systemic side effects. By acting mainly in the lungs, the rest of the body may experience less exposure to the treatment. This could improve patients’ quality of life during therapy.
In addition, this approach represents progress in the field of personalized medicine. Instead of attacking cancer in a general way, it seeks to modify the tumor’s specific environment.
This type of innovation also opens the door to future similar therapies for other respiratory diseases or types of cancer. The possibility of using inhalation as a delivery route for genetic therapies represents an important change in how treatments are administered.
The gene inhalable therapy is a promising experimental advance in lung cancer treatment. Although it is still under investigation, its early results show potential. This approach could offer more precise, less invasive, and better-tolerated treatments in the future, marking a new step in modern oncological medical innovation.
Referencia:
New Scientist/First ever inhalable gene therapy for cancer gets fast-tracked by FDA. Link
COMPARTE ESTE ARTICULO EN TUS REDES FAVORITAS:
Relacionado
Esta entrada también está disponible en:
Español
Discover more from Cerebro Digital
Subscribe to get the latest posts sent to your email.