The United States Food and Drug Administration has authorized an innovative system that uses a polymer activated by light to repair peripheral nerves without relying on traditional surgical stitches. The technology, developed by Tissium from academic research, introduces a less traumatic approach supported by recent scientific evidence.
How the Technology Works and What Exactly the FDA Approved
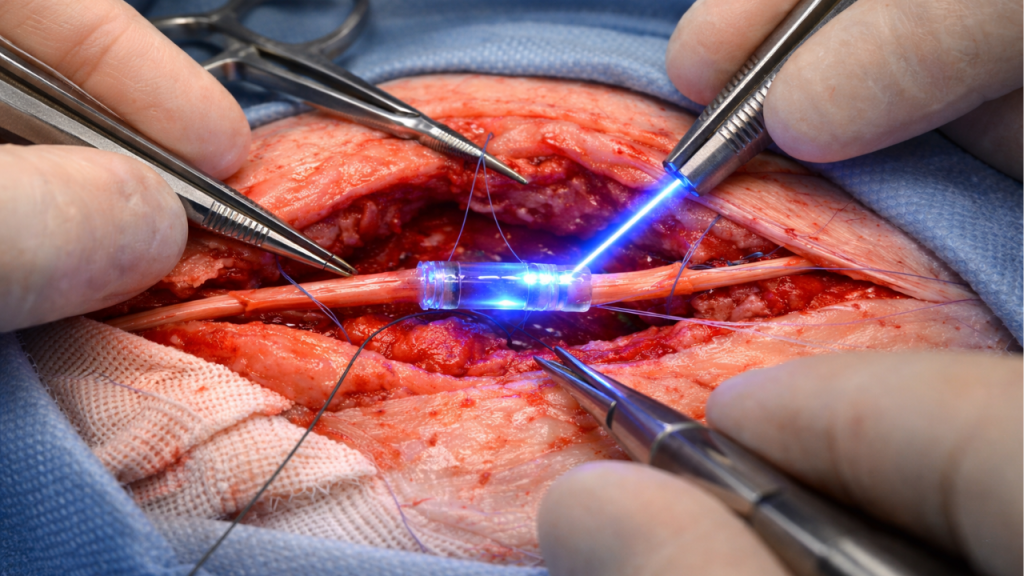
Repairing a severed peripheral nerve requires extremely precise alignment. Traditionally, surgeons perform neurorrhaphy with micro-sutures, a delicate procedure that requires a microscope and high technical skill. Although effective, this technique can generate localized compression and direct manipulation of the nerve tissue.
The system authorized by the FDA replaces those penetrating stitches with a coaptation method assisted by synthetic material. The procedure remains surgical: the surgeon approximates the nerve ends, but instead of passing sutures through them, applies a liquid biocompatible prepolymer around the junction area.
That material is activated by a controlled light source, triggering rapid polymerization. Within seconds, the compound solidifies and forms a flexible seal that keeps the nerve ends aligned. The goal is to achieve stability without repeatedly piercing the tissue.
According to the report published in Chemical & Engineering News, this is the first regulatory authorization for Tissium in this field. The device is designed to be bioabsorbable, meaning the polymer gradually degrades once the healing process has progressed.
It is important to emphasize that the approval does not eliminate surgery or magically accelerate regeneration. The innovation lies in joining the nerve using a polymer, not in altering the biology of the regenerative process.
What the Peer-Reviewed Scientific Study Demonstrated
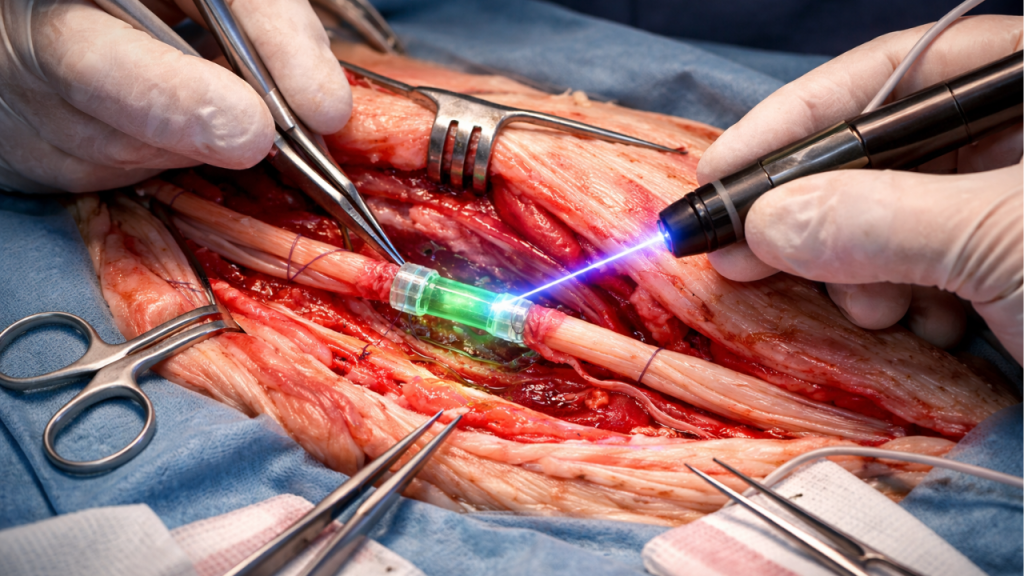
The technical foundation of this approach was evaluated in a study published in 2024 in Plastic and Reconstructive Surgery – Global Open. The research compared a polymer-assisted nerve repair system with conventional suture techniques.
The analysis focused on biomechanical parameters such as tensile strength, junction stability, and structural behavior under tension. The results showed that the system based on a polymer offered comparable strength to traditional neurorrhaphy.
A relevant finding was the more uniform distribution of force at the repair site. While sutures concentrate tension at specific points, the polymer creates more homogeneous contact, which could reduce localized microtrauma.
The study did not claim that the technique was superior in all clinical scenarios. However, it did support its mechanical feasibility and its potential as a safe alternative, providing scientific backing for the subsequent clinical development of the light-activated polymer.
Current Clinical Scope and Future Prospects
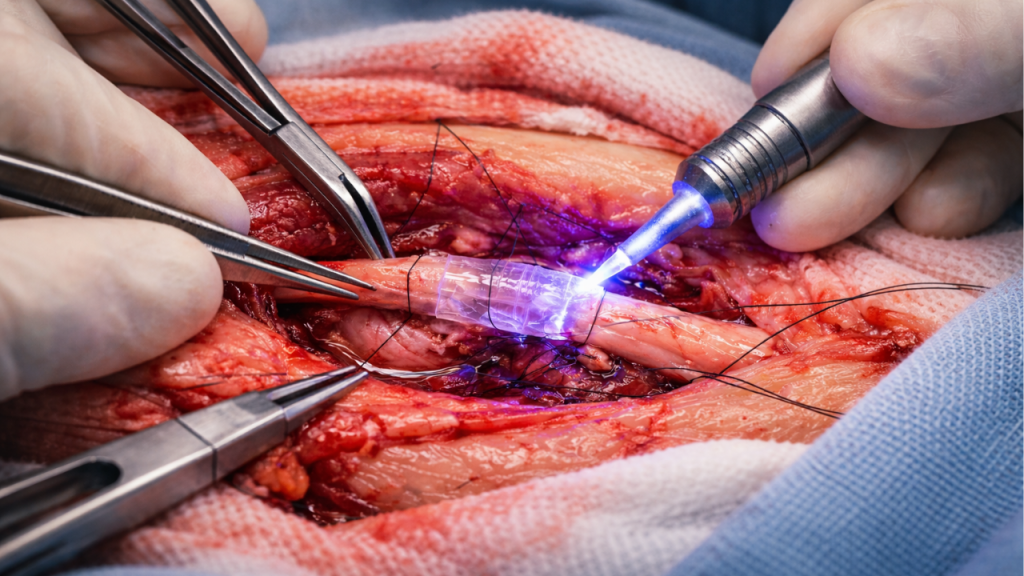
The FDA authorization focuses on peripheral nerve injuries in which the nerve ends can be approximated without excessive tension. It is not intended for all types of complex nerve damage.
Axonal regeneration still depends on the patient’s biology and may take months. The polymer acts as an initial structural support, facilitating stable alignment during the early stages of healing.
In addition, the bioabsorbable nature of the material prevents permanent implants from remaining in the body, potentially reducing long-term risks associated with foreign materials. This characteristic is one of the most notable advantages of the polymer-based technological platform.
The developing company has also indicated that the same polymer chemistry could be adapted for cardiovascular or gastrointestinal applications. If clinical studies continue to confirm safety and effectiveness, the approach could become a common alternative in reconstructive microsurgery.
The approval of the light-activated polymer represents a significant advance in peripheral nerve repair. Supported by peer-reviewed biomechanical evidence and regulatory authorization, the technology redefines how nerves are joined without replacing surgery. Its ultimate impact will depend on the long-term clinical outcomes observed in the coming years.
- Reference:
Plastic and Reconstructive Surgery – Global Open/Biomechanical Evaluation of an Atraumatic Polymer-assisted Peripheral Nerve Repair System Compared with Conventional Neurorrhaphy Techniques. Link
Chemical & Engineering News/FDA approves light-activated polymer for nerve repair without stitches. Link
COMPARTE ESTE ARTICULO EN TUS REDES FAVORITAS:
Relacionado
Esta entrada también está disponible en:
Español
Discover more from Cerebro Digital
Subscribe to get the latest posts sent to your email.








